Effect of xylo-oligosaccharides and β-glucan on gut microbiota and short chain fatty acid in diabetic rats
Abstract
Obesity and type 2 diabetes mellitus (T2DM) have an impact on an imbalance in the number of microbiota (dysbiosis) which can cause disruption of glucose metabolism by inhibiting short chain fatty acid (SCFA). SCFA is produced by intestinal microbial fermentation of indigestible dietary fibre and it has beneficial role in T2DM. Xylo-oligosaccharides (XOS) and β-glucans can improve the gut the composition of microbiota with prebiotic capabilities. The objective of the study was to investigate the effect of XOS and β-glucan on the Lactobacillus, Roseburia, and SCFA in diabetic rats. A total of 28 Wistar rats were divided into 4 groups including control (C), diabetic (D), diabetic+XOS (DX), and diabetic+β-glucan (DBG). D, DX, and DBG groups were provided with a high-fat diet (HFD) for 14 days before receiving an injection of streptozotocin (STZ) (45 mg/kg BW) and nicotinamide (110 mg/kg BW). DX received XOS (450 mg/200 g BW), and DBG received β-glucan (450 mg/200 g BW) for 28 days. The blood glucose levels at DX and DBG showed a significant difference compared with the D gro diabetic rats. XOS did not change the number, while β-glucan increased the number of Lactobacillus and Roseburia. The XOS and β-glucan supplementation significantly increased SCFA levels compared to diabetic rats. These results suggest that XOS can improve blood glucose although it does not change the number of Lactobacillus and Roseburia. Meanwhile, β-glucan increases blood glucose, Lactobacillus, Roseburia, and total SCFA in diabetic rats.
INTRODUCTION
The international diabetes federation states that the prevalence of diabetes mellitus worldwide was 9.3% in 2019. It is estimated to increase in 2030 to 10.2%, then in 2045, it is estimated to increase to 10.9% [1]. The results of a case study of diabetes mellitus in Indonesia show a trend of increasing from 6.9% in 2013 to 10.9% in 2018 [2]. Type 2 diabetes mellitus (T2DM) is characterized by hyperglycemia levels with random plasma glucose levels of ≥ 200 mg/dl [3]. There are several risk factors for type 2 diabetes related to changes in lifestyle such as lack of physical activity, high consumption of carbohydrates, and high fat leading to an increase in body mass index (BMI) which then develops into obesity [4].
Short chain fatty acid (SCFA) is the main metabolite of indigestible carbohydrate fermentation. The SCFAs most frequently found in the colon are acetate, propionate, and butyrate [5]. SCFAs produced through fiber fermentation are known to provide positive effects in preventing cardiometabolic disease including T2DM, obesity and cardiovascular disorders [6]. Furthermore, T2DM and obesity can influence an imbalance in the number of microbiota which is called as dysbiosis [7]. Dysbiosis can cause disruption of glucose metabolism by inhibiting the final products of SCFA metabolism which is specifically associated with a decrease in the number of butyrate-producing microbes. A decrease in the amount of butyrate results in modulating the immune response and it can cause low-grade inflammation which ultimately becomes one of the causes of T2DM [8].
Microbial composition and activity can be influenced by diet, such as the consumption of complex carbohydrates or prebiotics. Administration of prebiotics to experimental HFD-induced diabetic animals has been shown to improve the balance of microbiota [9]. Meanwhile, XOS consists of xylose oligomers which are linked through β-(1-4) linkages. XOS is a group of oligosaccharides which cannot be digested by humans, and it has a prebiotic effect. Prebiotics stimulate the production of activity of one or more bacteria in the colon (Bifidobacterium and Lactobacillus) by suppressing the activity of entero-decaying organisms and pathogens [10].
Consuming prebiotics is an effort to improve microbiota imbalance. β-glucan is a group of polysaccharides which have a prebiotic effect since they can modulate the intestinal microbiota. Consumption of β-glucan from oats or barley increases the growth of Roseburia population producing SCFAs such as propionate and butyrate [11]. Research shows that administration of oats and barley containing β-glucan stimulates the growth of Lactobacillus and Bifidobacteria, which can help to inhibit dysbiosis [12].
Based on the description above, XOS and β-glucan have potential effects on metabolic parameters as well as gut microbiota. Therefore, this study aimed to determine the effect of administering XOS and β-glucan on the number of Lactobacillus, Roseburia and SCFA composition in diabetic Wistar rats.
MATERIALS AND METHODS
Animals
This study was a true experimental post-test only with a randomized controlled group design. In this study, 28 male Wistar rats weighing 160 to 220 g aged 8 weeks were used which were divided into 4 groups such as control (C), diabetic (D), diabetic+XOS (DX), and diabetic+β-glucan (DBG). Each rat was acclimatized for 7 days in a cage, humidity ranges from 70-75% while temperature ranges from 25-28⁰C, the light settings were set to 12 am and 12 pm at the Experimental Animal Laboratory of the Centre for Food and Nutrition Studies, Gadjah Mada University, Yogyakarta, Indonesia. This research was conducted from December 2022 to February 2023. All of the rats were adjusted to room temperature, and they were given Comfeed AD II food standard and water ad libitum during the study. After the acclimatization period, the initial body weight of the rats was recorded. The control group was only given standard Comfeed AD II feed and treated as healthy rats. Groups D, DX, and DBG were given high fat diet (HFD) for 14 days and treated with streptozotocin (STZ) 45 mg/kg BW and nicotinamide (NA) 110 mg/kg BW. The DX group received XOS at 450 mg/200g BW. The DBG group received at 450 mg β-glucan/200g BW. XOS and β-glucan were given for 28 days, respectively. XOS was purchased from Shandong Longlive Biotechnology (Shandong, China) and β-glucan from Lake Avenue Nutrition (Madre Labs, Laguna Canyon Rd). At the end of the study, all of the rats were dissected, and their cecum was taken, then placed in a falcon tube. The cecum was weighed 500 mg and then put into a microtube. After collection, the cecum was transported on ice to the Biochemistry Laboratory at the Gadjah Mada University Faculty of Medicine, Public Health, and Nursing, where it was kept at -20°C until it was analyzed.
Animal facilities, their management, and handling during the experiment were done in compliance with the Guidelines for Care and Use of Laboratory Animals of CNFS Gadjah Mada University and were approved by the Research Ethics Committee of the Faculty of Medicine, Diponegoro University (No.120/EC/H/FK-UNDIP/X/2022).
Blood glucose analysis
Blood glucose levels were checked 3 times such as a) at starting of the study, b) 3 days after the STZ+NA treatment to ensure that the rats were hyperglycemic, and c) at the end of the study. All rats were fasted 8-10 h before taking blood. 2 ml of blood was taken through the plexus retroorbital, then put in a tube containing EDTA anticoagulant. The samples were centrifuged at 3000 rpm for 15 minutes, then the serum was collected. The analysis was carried out using the Enzymatic colorimetric test using GOD-PAP method. Rats are declared diabetic when their blood glucose levels were greater than 250 mg/dl [13].
SCFA analysis
SCFA components were analyzed using Gas Chromatography Mass Spectrophotometry. Briefly, 10 mg of cecum was taken into a 1.5 ml tube and added 400 μL of water andhomogenized by vortexing. Then, it was centrifuged at 1400 rpm for 10 min. The supernatants were transferred into 1.5 mL tubes. Then, the supernatants were diluted 1:5 with water for a total volume of 50 μL using a 1.5 mL tube [14]. In addition, the samples were analyzed by Shimadzhu GC-2010 Plus. SCFA levels were analyzed at the Food Technology and Agricultural Products Testing Laboratory, Gadjah Mada University, Yogyakarta, Indonesia.
Cecum bacterial DNA extraction
DNA od cecum samples were extracted using Favor PrepTM DNA isolation kit (FavorGen Biotech, Taiwan, China) following the manufacturer’s instruction. Total DNA concentration was calculated by using nanodrop (Maestrogen Inc).
Standard curves creation
The Lactobacillus inoculum at concentration of 109 CFU/g was obtained from the Laboratory of the Center for Food and Nutrition Studies Gadjah Mada University and it was gradually diluted to 102 CFU/g. After dilution, it was centrifuged at 5000 rpm for 5 min at 4°C until a precipitate was formed. After that, the samples were rinsed with 10 mL of 0.9% NaCl. Then the centrifugation and vortexing was conducted again until a precipitate was created.
G-blocks Gene Fragment Roseburia with a concentration of 500 ng which had been designed and ordered at (Genetica Science Indonesia). The dilution process began with spin down for 15 seconds to ensure the material is at the bottom of the tube, 50 μ/L nuclease free water (NFW) was added until it reached a final concentration of 10 ng/ μL. Then, it was vortexed briefly, incubated at 50⁰ C for 20 min, and again vortexed briefly. Finally, 1 μ/L was taken, put it in a new 2 ml tube which contained 99 μ/L NFW, and then conducted gradual dilution until 102 CFU/g.
The logarithmic concentration of bacteria inoculum dilutions and G-Blocks were plotted against a threshold number in order to produce a standard curve. The standard curve obtained was as follows: Lactobacillus: Y = -3.3789x + 47.084 (R2 = 0.9926) and Roseburia: Y= -3.468x + 44.944 (R2 = 0.9782). The plotting results in the dilution range showed a linear relationship in which the line detected is a dilution of 1x102 - 1x109 copies/μL.
Quantification of bacterial by RT-PCR
The primers used to detect Roseburia were based on the 16srRNA gene sequence taken from the BLAST database desaigned an ordered from Genetika Science Indonesia. The primer sequences for Roseburia were forward-GCCACATTGGGACTGAGACA and reverse-GCTGCTGGCACGTATTTAGC. Meanwhile, the Lactobacillus primer sequences as in previous research, was primers forward-AGCCAGTAGGGAATCTTCCA and reverse-CACCGCTACACATGGAG [15]. The mixture composition in the PCR tube contains 10 μl of ExcelTaq 2x Fast Q-PCR SYBR master mix (SMOBIO Technology Inc), 1 μl of reverse primer and 1 μl of forward primer each, the amount of bacterial DNA samples adjusted to the nano drop results, the amount of nuclease free adjusted the bacterial DNA samples to reach a total volume 20 μl. Furthermore, the mixture in the PCR tube was analyzed using Bio Rad CFX96 Real-Time. The PCR temperature setting was as follows: 95⁰C for 10 min, followed by 40 cycles for 15 seconds at 95⁰C and 1 min at 60⁰C for Lactobacillus while temperature settings for Roseburia was: 95⁰C for 10 min, followed by 40 cycles for 15 seconds at 95⁰C and 30 seconds at 54⁰C. After the running process, melting curve analysis was carried out to confirm to ensure the accuracy of the RT-PCR data. The standard curve of the bacterial strain was used to calculate the cycle thresholds of bacterial DNA. Log CFU/gr was used to express the results.
Statistical analysis
SPSS 23 software (IBM/SPSS Inc.) was used to perform statistical analysis. Meanwhile, the Shapiro-Wilk normality test was used to analyze the research data. Post-intervention data were normally distributed using one way ANOVA on the effect of blood glucose, Lactobacillus and SCFA levels. Because the Roseburia data were not normally distributed, Kruskal Wallis analysis was used as a non-parametric test. The P value < 0.05 was considered significant, and all results were presented as mean ± SD.
RESULTS
Effects of XOS and β-glucan on blood glucose levels in diabetic rats
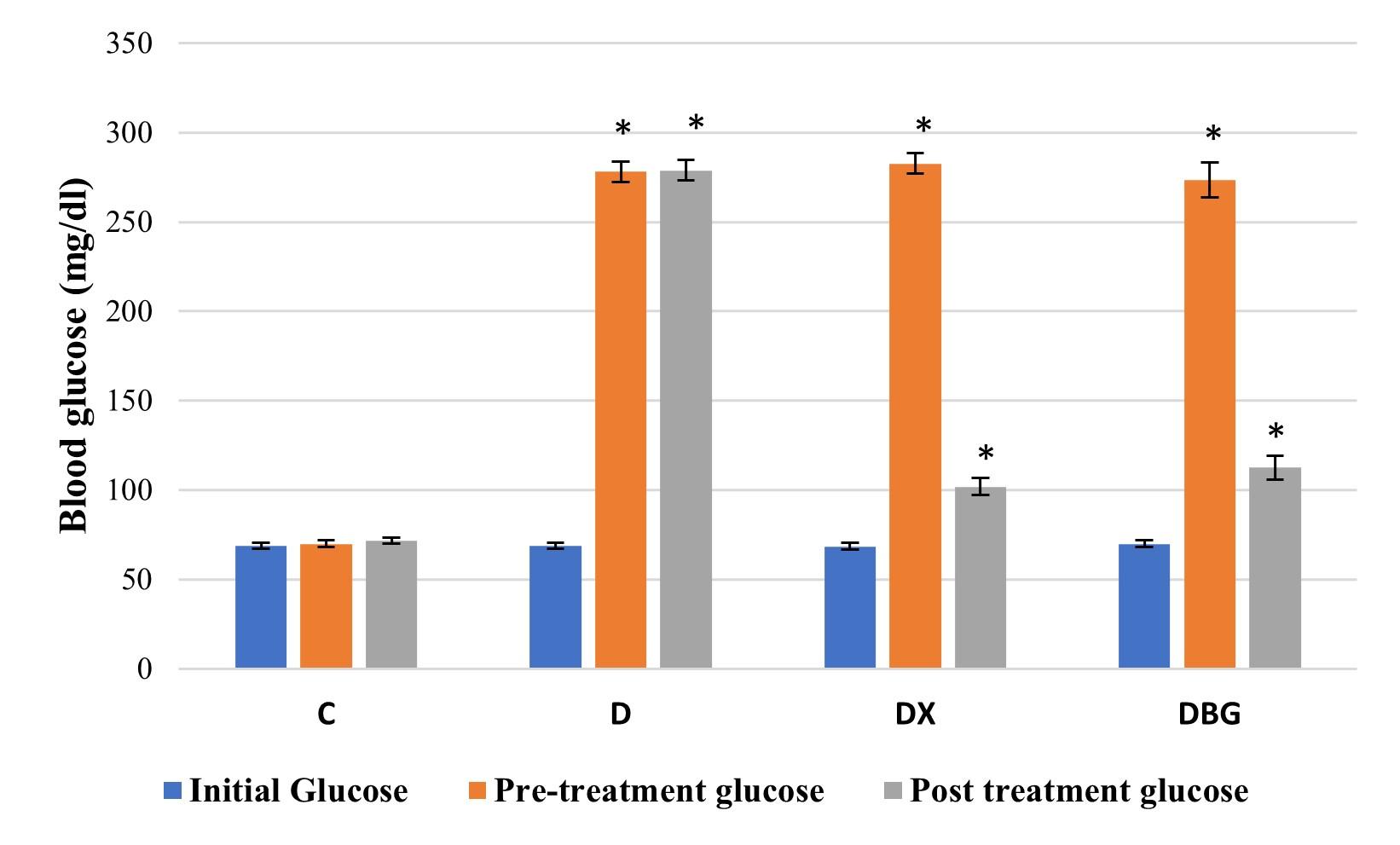
Administration of HFD and STZ showed an increase in blood glucose levels in rats indicating that the rats had hyperglycemia (Table 1). Supplementation with XOS and β-glucan 450 mg/200 g BW in DX and DBG group showed reduced blood glucose levels, respectively (Figure 1 and Table 1). The Shapiro-Wilk normality test on blood glucose showed significant difference in all groups, indicating that the data is normally distributed (Figure 1 and Table 1). The one-way ANOVA test showed a p value = 0.000 (p<0.05), indicating that there is a significant difference in blood glucose levels across all treatment groups. LSD post-hoc test was used to determine which groups were statistically different. The results in Figure 1 showed significant differences in blood glucose levels in D, DX and DBG groups against the C group.
Table 1. Effect of XOS and β-Glucan on blood glucose of HFD-STZ-induced diabetic rats.
Effects of XOS and β-glucan on Lactobacillus and Roseburia in diabetic rats
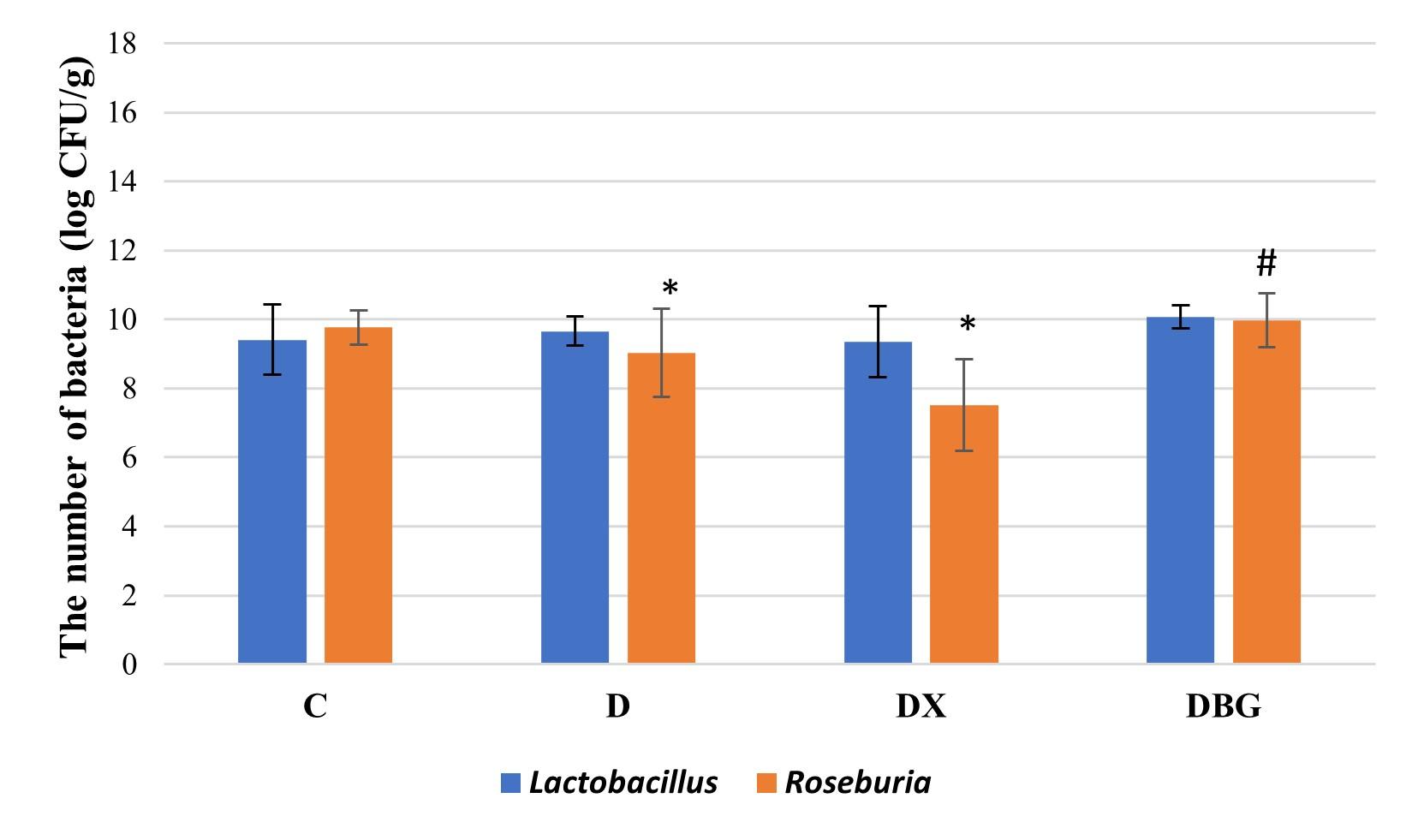
In the cecum, the number of Lactobacillus in diabetic group was higher than in control group (Figure 2 and Table 2). In contrast, the number of Lactobacillus in the cecum of DX group was decreased than baseline. Compared to control group and DX group, the cecum of the DBG group contained more of Lactobacillus. According to Shapiro Wilk test for the Lactobacillus, the outcomes for all groups were significant (p>0.05), indicating that the data is of normal distribution (Figure 2 and Table 2). The one-way ANOVA test showed a p = 0.313 (p>0.05), indicating there is no significant difference in the number of Lactobacillus among all treatment groups (Figure 2 and Table 2). In addition, the number of Roseburia in diabetic group decreased compared to the control group (Figure 2 and Table 2). It means that rats with diabetes mellitus had dysbiosis. However, in the DX group total Roseburia was slightly decrease than control and diabetic groups. Interestingly, the number of Roseburia in DBG group is greater than C, D, DX. The Shapiro-Wilk normality test showed that Roseburia were not normally distributed (p<0.05) (Figure 2). The Kruskal Wallis test showed a p = 0.021 (p<0.05), indicating there was significant difference in the number of Roseburia. LSD post-hoc test was used to determine which groups were statistically different. The results in Figure 2 showed significant differences in the number of Roseburia between diabetic and C groups as well as significant differences between DBG and diabetic groups.
Table 2. The number of Lactobacillus and Roseburia after the intervention.
Effects of XOS and β-glucan on SCFA in diabetic rats
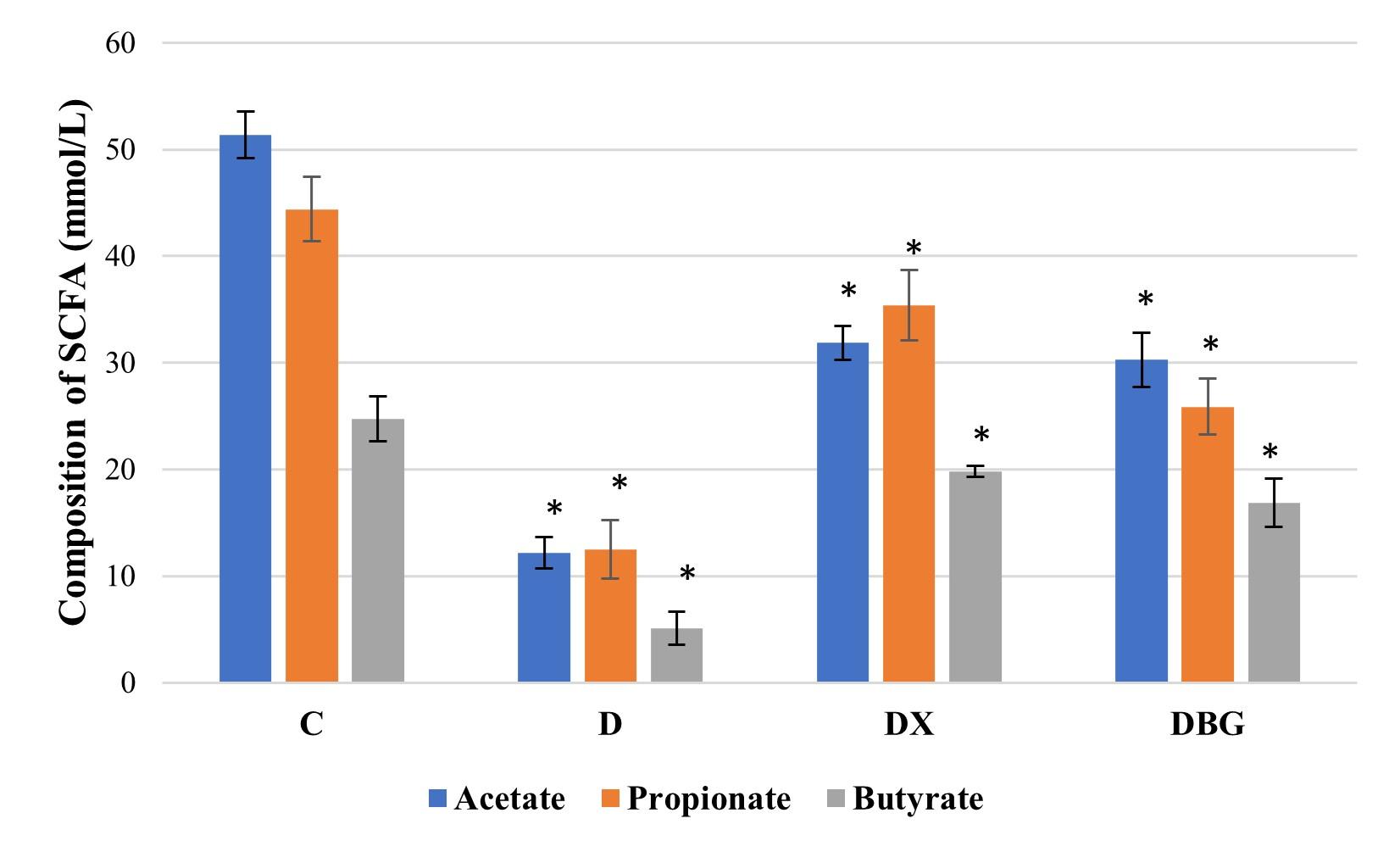
SCFA in the cecum were measured to verify the presence of the final products after XOS and β-glucan administration. In the DX and DBG groups, propionate, acetate, and butyrate levels were substantially higher after receiving XOS and β-glucan treatment in diabetic rats (Figure 3 and Table 3). The distribution of SCFA in the diabetic group decreased compared to the control group. In the DX group, the amount of propionate is found to be higher while in the DBG group the amount of acetate is higher. The Shapiro-Wilk normality test for on SCFA significance in all group (p>0.05), indicating that the data was of normal distribution (Figure 3). The one-way ANOVA test showed a p = 0.000 (p<0.05), indicating there was a significant difference in SCFA across all treatment groups. LSD post-hoc test was used to determine which groups were statistically different. The results showed significant differences in the SCFA components between groups (Figure 3 and Table 3).
Table 3. SCFAs level after intervention.
DISCUSSION
The characteristic of diabetes mellitus is chronic hyperglycemia caused by lack of insulin secretion, and lack of insulin action on target tissues, or both [16]. Recently, great attention has been paid to derivative products made from natural ingredients intended to help people suffering from diabetes. Our study showed that administration of XOS and β-glucan at a dose 450 mg/200 g BW for 28 days showed a beneficial effect in reducing blood glucose levels in rats with T2DM which indicates anti-hyperglycemic outcomes.
Nowadays, various experimental animal models have been developed to study diabetes. For a decade, STZ, a natural glucosamine-nitrosourea, has been used to induce diabetes in rats. After the STZ treatment, pancreatic β cells are destroyed due to the alkylation process of the diabetogenic agent STZ, causing decreased insulin production and uncontrolled plasma glucose levels in diabetic rats [13]. Administration of low doses of STZ and HFD are often used to induce diabetes in rats [17]. The characteristics of diabetic rat include abdominal obesity, insulin resistance, hyperglycemia, hyperinsulinemia, and dyslipidemia, similar to human condition. These conditions are suitable for studying the therapeutic effects on diabetes in our experimental study.
The biological properties of XOS have been demonstrated by numerous reports [18]. Furthermore, commercial administration of XOS to STZ-induced diabetic rat for 5 weeks of intervention showed improvement in several diseases including inhibiting of increases in glucose, cholesterol and triglyceride concentrations. XOS can significantly increase fecal content as well as cecal acetate concentration in STZ-induced diabetic rats [19]. Similarly, our results showed beneficial effects on blood glucose after XOS administration in STZ-induced and HFD-fed in rats. It is in line with previous clinical studies show that administration of XOS (4g/day) for 8 weeks has advantage effects on blood glucose, HbA1C, fructosamine, and lipid profile in diabetic patients [20]. These data contribute to the confirmation that XOS has hypoglycemic and hypolipidemic effects as well as amelioration of insulin resistance. XOS has been reported in various studies to have benefits in diabetes both in vivo and clinically, but the underlying mechanisms are still unclear [21].
The results of our studies showed that administration XOS at 450 mg/ 200 g BW does not increase the number of Lactobacillus. It is in line with previous study that XOS supplementation does not increase Lactobacillus [22]. Lactobacillus in the diabetic rat group increased compared to the control group. The results of research in human show that the gut microbiota associated with obesity has increased levels of Lactobacillus [23]. Another study shown in Southern China populations shows that T2DM cause increased numbers of L. Bulgaricum, L, Rhamnosus and L. acidophilus compared with control group. In type 2 diabetes mellitus, related lactic acid bacteria such as Enterococcus, Streptococcus and Lactobacillus are decreased due to XOS [24]. In addition, this study shows that the number of Roseburia does not increase with XOS supplementation.
In vitro and in vivo studies show that SCFAs stimulate the secretion of glucagon-like-peptide-1 (GLP-1) and peptide YY (PYY) in the intestine by activating GPR41 and GPR43 [25]. Moreover, GLP-1 and PYY have a very good role in increasing satiety through the gut-brain-axis. This process can indirectly reduce appetite and food intake so that it inhibits weight gain, which is considered a risk factor for T2DM. In addition, SCFAs can regulate blood glucose through increasing GLP-1-mediated insulin secretion [26]. Additionally, acetate and butyrate affect skeletal muscle glucose metabolism in an AMPK-dependent manner that in turn increases glucose uptake (via GLUT4) and glycogen storage via a GPR41/GPR43-mediated mechanism. SCFA indirectly influences muscle insulin sensitivity and glucose metabolism through increasing gut-derived PYY and GLP‑1 levels so that it affects skeletal muscle insulin action and glucose absorption that contribute to increased muscle insulin and glucose sensitivity [27].
The result of this study shows that XOS is able to increase the amount of SCFA, especially propionate. Propionate enhances glucose-stimulated insulin release by pancreatic β-cell in vivo via activating FFAR2 and protecting β cells from apoptotic stimuli [28]. Another advantage of propionate in terms of health benefits is due to its anti-lipogenic, cholesterol-lowering, anti-inflammatory, and anti-cancer effects [29]. Furthermore, in the β-glucan group, we find higher acetate. It has been suggested that acetate plays a direct role in central appetite regulation [30]. Based on the results of previous research, it was shown that giving high fat to diabetic rats can reduce total SCFA; especially, butyrate. Total butyrate is associated with reduced glucose, insulin resistance and body weight in diabetic rats fed a high-fat diet. Butyrate deficiency can alter glucose metabolism and worsen the pathogenesis of diabetes by inducing inflammation and oxidative stress [31].
β-glucan is a soluble fiber polysaccharide with a mixture of β-(1-3) and β-(1-4) glycosidic bonds. β-glucan has health benefits, including lowering blood glucose and cholesterol in obesity and cardiovascular disease. The study shows that low oat β-glucan intake significantly lowered blood glucose and insulin levels. The positive hypoglycemic effect of β-glucan is due to its viscosity effect [32]. The interaction of β-glucan with intestinal mucus is considered as another potential mechanism of cereal β-glucan in the regulation of postprandial blood glucose and insulin levels [33]. Moreover, studies report that oat β-glucan particles, which are part of the cell wall, exist in a natural tissue-like structure which can encapsulate protein and starch in order to form a complex cell matrix in the cell wall. This matrix can reduce access enzyme which could slow down leads to reduced starch digestion and decreases postprandial glycemic response [34].
Based on studies, the indigestible form of β-glucan found in cereals can pass through the digestive tract. As a result, it can serve as a substrate for microbial fermentation and specifically promotes the growth and activity of a number of small beneficial microorganism, such as Bifidobacteria and Lactobacillus, which occur during the formation of SCFA [35]. The prebiotic impact of cereal β-glucan has now been linked to the viscosity of β-glucan and it has been shown to be influenced by the volume of non-fermentable polysaccharides in the colon of rats [36]. According to previous studies, the effects of oat β-glucan on intestinal flora depend on molecular weight and dose. The better the effect so that the lower the molecular weight and the larger the dose. In addition, recent studies have shown that administration of cereals β-glucan increase the number of Lactobacillus in rats [37].
CONCLUSIONS
Based on this study, it can be concluded that XOS can improve blood glucose and increase SCFA in diabetic rats. However, XOS did not increase the number of Lactobacillus and Roseburia. It is important to be paid more attention to increase the dose and duration of the treatment by using XOS and environmental factors, such as measuring the pH in cecum. In conclusion, β-glucan can improve the blood glucose in diabetic rats so that it can increase the number of Lactobacillus and Roseburia as well as SCFA.
ACKNOWLEDGMENT
The authors would like to thank Mr. Yulianto for his assistance in the care and maintenance of laboratory animals. The researcher also would like to thank Mr. Rio Jati Kusuma, Ms. Yuenleni, and Ms. Ida Darojatun for their assistance in DNA identification process.
AUTHOR CONTRIBUTIONS
IEZ, the main researcher on the study, had designed and conceived, written the manuscript, and performed data collection. AZJ and EM checked the text and contributed suggestions on its interpretation and data analysis. Meanwhile, MS and AP were reviewers of the manuscript. The final version of the manuscript was approved by all authors.
CONFLICTS OF INTEREST
There is no conflict of interest among the authors.
References
- [1]Saeedi P, Petershon I, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Research and Clinical Practice. 2019; 157: 1–10.
- [2]Indonesia KKR. Hasil Utama Riskesdas 2018. 2018: 1-47.
- [3]Association AD. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2020; 33:2
- [4]Wu Y, Ding Y, et al. Risk factors contributing to type 2 diabetes and recent advances in the treatment and prevention. Int. J. Med. Sci.2014; 11: 1185-1200.
- [5]Morrison D. J and Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. 2016; 7: 189-200.
- [6]Nogal A, Valdes A. M et al. The role of short-chain fatty acids in the interplay between gut microbiota and diet in cardio-metabolic health. Gut Microbes. 2021; 13: 1-24.
- [7]Liu Y, and Lou X. Type 2 diabetes mellitus-related environmental factors and the gut microbiota: Emerging evidence and challenges. Clinics. 2020; 75: 1-7.
- [8]Ahmad A, Yang W, et al. Analysis of gut microbiota of obese individuals with type 2 diabetes and healthy individuals. PLoS One. 2019; 14 :1-16..
- [9]Bezirtzoglou E, Stavropoulou E, et al. Maintaining digestive health in diabetes: The role of the gut microbiome and the challenge of functional foods. Microorganisms. 2021; 9: 1-24.
- [10]Mhetras N, Mapre V et al. Xylooligosaccharides (XOS) as Emerging Prebiotics: Its Production from Lignocellulosic Material. Adv. Microbiol. 2019; 9: 14-20.
- [11]Fehlbaum S, Prudence K, et al. In vitro fermentation of selected prebiotics and their effects on the composition and activity of the adult gut microbiota. Int. J. Mol. Sci. 2019; 19: 1-16.
- [12]Shoukat M and Sorrentino A. Cereal β-glucan: a promising prebiotic polysaccharide and its impact on the gut health. Int. J. Food Sci. Technol. 2021; 56: 2088–2097.
- [13]Ghasemi A, Khalifi S, et al. Streptozotocin-nicotinamide-induced rat model of type 2 diabetes (review). Acta Physiologica Hungaria. 2014; 101: 408-420
- [14]Hoving LR, Heijink M, et al. Gc-ms analysis of short-chain fatty acids in feces, cecum and blood samples. Clinical Metabolomics: Methods and Protocols, Methods in Molecular Biology. 2018;1730:247-56.
- [15]Zimmermann MB, Cassard C, et al. The effects of iron fortification on the gut microbiota in african children: A randomized controlled trial in coˆ te d’ivoire. Am J Clin Nutr. 2010; 92 : 1406–1415.
- [16]Association AM. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014; 37: 81–90.
- [17]Barrière DA, Noll C, et al. Combination of high-fat/high-fructose diet and low-dose streptozotocin to model long-term type-2 diabetes complications Scientific Reports. 2018; 8 : 1-17.
- [18]Kumar GP, Pushpa A, et al. A review on xylooligosaccharides. Int. Res. J. Pharm. 2012; 3: 71–74.
- [19]Gobinath D, Madhu AN, et al. Beneficial effect of xylo-oligosaccharides and fructo-oligosaccharides in streptozotocin-induced diabetic rats. British. J. Nutr. 2010; 104: 40–47.
- [20]Sheu WHH., Lee IT, et al. Effects of xylooligosaccharides in type 2 diabetes mellitus. J. Nutr. Sci. Vitaminol. 2008; 54: 396–401.
- [21]Khat-udomkiri N, Toejing P, et al. Antihyperglycemic effect of rice husk derived xylooligosaccharides in high-fat diet and low-dose streptozotocin-induced type 2 diabetic rat model. Food Sci. Nutr. 2020; 8: 428–444.
- [22]Finegold SM, Li Z, et al. Xylooligosaccharide increases bifidobacteria but not lactobacilli in human gut microbiota. Food and Function. 2014; 5: 436–445.
- [23]Armougom F, Henry M, et al. Monitoring bacterial community of human gut microbiota reveals an increase in Lactobacillus in obese patients and Methanogens in anorexic patients. PLoS One. 2009; 4: 1-8.
- [24]Marlene R, Dworzak S, et al. Abundance and Diversity of Microbiota in Type 2 Diabetes and Obesity. J. Diabetes Metab. 2013; 4: 1-8.
- [25]He J, Zhang P, et al. Short-chain fatty acids and their association with signalling pathways in inflammation, glucose and lipid metabolism. Int. J. Mol. Sci. 2020; 21: 1-16.
- [26]Salamone D, Rivellese AA, et al. The relationship between gut microbiota, short-chain fatty acids and type 2 diabetes mellitus: the possible role of dietary fibre. Acta Diabetol. 2021; 58: 1131–1138.
- [27]Canfora EE, Jocken JW, et al. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015; 11: 577–591.
- [28]Pingitore A, Chambers ES, et al. The diet-derived short chain fatty acid propionate improves beta-cell function in humans and stimulates insulin secretion from human islets in vitro. Diabetes, Obes. Metab. 2016; 19: 257-265
- [29]Hosseini E, Grootaert C, et al. Propionate as a health-promoting microbial metabolite in the human gut. Nutr. Rev.2011; 69: 245–258.
- [30]Frost G, Sleeth ML,et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 2014; 5: 1–11.
- [31]Noureldein MH, Bitar S, et al. Butyrate modulates diabetes-linked gut dysbiosis: Epigenetic an mechanistic modifications. Journal of Molecular Endrocinology. 2020;64:29-42.
- [32]Bai J, Ren Y, et al. Physiological functionalities and mechanisms of β-glucans. Trends Food Sci. Technol. 2019; 88: 57–66.
- [33]Mackie A, Rigby N, et al. Increasing dietary oat fibre decreases the permeability of intestinal mucus. J Funct Foods. 2016: 418-427.
- [34]Hu H, Lin H, et al. Impact of Native Form Oat β-Glucan on the Physical and Starch Digestive Properties of Whole Oat Bread. Foods. 2022; 11: 1–11
- [35]Havrlentová M, Petrulakova Z, et al. Cereal β-glucans and their significance for the preparation of functional foods – a review. Czech Journal Food Science. 2011;29:1-14.
- [36]Monro JA, Paturi G, et al. Prebiotic effects of fermentable carbohydrate polymers may be modulated by faecal bulking of non-fermentable polysaccharides in the large bowel of rats. Int. J. Food Sci. Technol. 2012; 47: 968–976.
- [37]Shen RL, Dang XY, et al. Effects of oat β-glucan and barley β-glucan on fecal characteristics, intestinal microflora, and intestinal bacterial metabolites in rats. J. Agric. Food Chem, 2012; 60: 11301–11308.