Effectiveness of a gluten-free diet on the autoantibody frequency of endocrine and neuron disorders in patients with celiac disease
Abstract
Several possible associations between different types of autoimmune diseases have been reported, therefore this study aimed to investigate the relationship between celiac disease (CD) and some related autoimmune diseases by determining the effect of gluten on severity of these diseases. Two hundred blood samples were collected, 50 samples were withdrawn from healthy donors as a control group, 75 were collected from newly diagnosed (ND) CD patients, while the remaining blood samples (75) were obtained from the same patients after 4 months of gluten free diet (GFD) regime. Sera were separated to measure the level of anti-tissue transglutaminase IgA (tTg-IgA), anti-pituitary IgG, anti- thyroid peroxidase IgG (TPO- IgG), anti-islet IgG and anti-ganglioside IgG (IU/ml) by enzyme linked immunosorbent assay (ELISA). The results recorded high significant elevation (p>0.0001) in the concentrations of all autoantibodies of both ND and GFD groups compared with their concentrations in the control group. Furthermore, after 4 months of GFD, concentrations of all autoantibodies revealed highly significant decreases (p>0.0001) as compared with ND. Regarding the correlation of all studied autoantibodies with anti tTG-IgA, only anti-ganglioside IgG levels were weakly correlated in pre-GFD treatment patients, meanwhile anti-ganglioside IgG and anti-pituitary IgG were also recorded weak correlations in post- GFD of CD patients. This study concluded that GFD helps to reduce the severity of endocrine and neuron autoimmune disorders in addition to CD, which have been proved by decreasing in the level of their diagnostic autoantibodies.
INTRODUCTION
Celiac disease (CD) is an autoimmune disease that cause intestinal chronic inflammation and it is one of the most common cause of food intolerances in the world, with a prevalence ranged from 0.5%to 2.0% in the general population with minor regional differences [1]. Early CD diagnosis and treatment based on a gluten free diet (GFD), which help to avoid a number of problems such as the emergence of other autoimmune illnesses, osteoporosis, or cancer (especially T-cell lymphoma) [2, 3].
Gluten is an ethanol soluble protein and constituted from many immunogenic peptides. These components have the ability to resist digestive enzymes to trigger the inflammatory process which could be harmful with a long-standing complication [4]. Gluten is found in some grains, including wheat, barley and rye [4].
First-degree relatives of CD patients, those with type 1 diabetes mellitus (T1DM), autoimmune thyroiditis, autoimmune hepatitis, selective IgA deficiency, and other genetic disorders are among people who are at high risk for CD [5]. Compared to 0.5% of the general population, patients with T1DM had a prevalence of CD ranging from 4.4 to 11.1% [6]. T1DM is associated with other organ specific antibody mainly thyroid, adrenal glands and CD, which reinforces the necessity of CD and thyroid screening in T1DM subjects [7].
In a recent investigation, insulin autoantibodies were revealed to be the most often occurring islets autoantibodies, followed by glutamic acid decarboxylase and zinc transporter 8. The quantity of antibodies present may affect future risk of CD but not future risk of Hashimoto's disease [8]. The relationship of two illnesses may have significant clinical ramifications. One hypothesis refers to the cross reaction of anti-tissue transglutaminase IgA (tTg-IgA) with thyroid tissue [9]. Clinically identifying CD may be challenging in people with hypothyroidism since the amount of circulating thyroid hormone is reduced, which may result in fluid retention or less severe diarrhea or weight loss. Additionally, CD decreases the tiny intestinal surface area that can absorb nutrients, which prevents hypothyroid patients from responding to oral thyroid replacement medication. Additionally, decreased absorption and an accelerated transit rate in hyperthyroidism may be to blame for an apparent inability to respond to a GFD in CD patients. The use GFD is controversial; some studies found no benefit, while others found that utilizing a GFD can normalize thyroid function and decrease thyroxine dosage with recovery from autoimmune thyroid disease that is either subclinical or clinical [10].
Gluten sensitivity can be demonstrated with several neurological dysfunctions including neuropathy, encephalopathy, and ataxia with or without associated CD. Gluten sensitivity also correlate with central nervous system hyperexcitability which is often associated with refractory CD [11].
The purpose of this study was to determine whether a GFD has a significant effect on the levels of autoantibodies resulting from an immune disorder in some parts of endocrine system (anti-pituitary IgG, TPO- IgG and anti-islet IgG) and nervous system (anti-ganglioside IgG) that may be associated with CD. Furthermore, the present study investigated the correlation of these autoantibodies with tTG- IgG having pre and post GFD treatment.
MATERIALS AND METHODS
Study participants
This longitudinal clinical intervention study was performed where enrolled patients were diagnosed clinically in Raparin Children’s hospital and Rizgary Teaching hospital in Erbil city, Iraq. Serologically, CD patients identified according to tTG IgA test with a weak positive level of tTG IgA were excluded. The study included the withdrawing of 150 blood samples from CD patients, which were divided as 75 blood samples from ND of CD patients and 75 samples from the same patients after 4 months of GFD treatment. In addition, 50 blood samples were collected from apparently healthy persons. The duration of the study was 13 months (from July 2021 to October 2022).
The study proposal was confirmed by the ethics committee of Erbil Technical Health and Medical College, Erbil Polytechnic University, Iraq, according to the document number, REC 2/84.
Sample collection and laboratory measurements
Firstly, 5 ml of blood was collected from all participants then sera were separated and kept at - 20 ºC until used. Serum levels of anti-tTG IgA, anti-pituitary IgG, anti-TPO IgG, anti-islet IgG and anti-ganglioside IgG antibodies were measured by ELISA technique using commercially available kits (MyBiosource- California, USA) where standard and sera were added to microtitration plate to detect each antibody in a separate well. A plate was read at 450nm using a plate reader (BIO-TEK - USA) after several steps of incubation and washing, these procedures were repeated to determine the concentration of each antibody under study.
Statistical analysis
All obtained data were analyzed using GraphPad Prism program version 8. Normality test, Mann-Whitney test, Wilcoxon test in addition to spearman’s correlation test were performed to determine the significance alterations. The results were recorded as mean ± standard deviation (SD) and significant differences were reported when the P value was less than 0.05.
RESULTS
Serum levels of different autoantibodies in CD patients
Results elucidated in Table 1 revealed that the serum level of anti-tTG IgA (116.6 ± 54.26 IU/ml), anti-pituitary IgG (116.0±93.62 IU/ml), anti-TPO IgG (32.90±34.11 IU/ml), anti-islet IgG (50.54±48.07 IU/ml) and anti-ganglioside IgG (10.72±11.27 IU/ml) were significantly higher (P < 0.05) in ND group of CD patients than control (3.170 ± 1.637, 19.11±16.49, 10.38±6.597, 3.546 ± 2.300 and 4.942 ± 3.045 IU/ml), respectively.
Table 1. Comparison of autoantibodies between ND group of CD patients and control.
Effect of GFD treatment on autoantibodies in CD patients
Serum anti-tTG IgA (67.03±28.78IU/ml), anti-pituitary IgG (85.75±71.93 IU/ml), anti-TPO IgG (21.86±23.80 IU/ml), anti-islet IgG (40.76±39.96 IU/ml) and anti-ganglioside IgG (8.370±8.346 IU/ml) levels were significantly higher (P < 0.05) in the CD patients under GFD group as compared with control (3.170 ± 1.637, 19.11±16.49, 10.38±6.597, 3.546 ± 2.300 and 4.942 ± 3.045 IU/ml, respectively) (Table 2).
A comparison between CD patients, pre and post treatment for 4 months with GFD, was conducted with respect to the concentrations of autoantibodies (anti-tTG IgA, anti-Pituitary IgG, anti-TPO IgG, anti-Islet IgG and anti-ganglioside IgG). The result exhibited that the mean levels of all tested autoantibodies in CD patients were significantly (p < 0.0001) decreased after 4 months of GFD regime compared with ND CD patients before starting the regime (116.6±54.26 vs 67.03±28.78; 116.0±93.62 vs 85.75±71.93; 32.90±34.11 vs 21.86±23.80; 50.54±48.07 vs 40.76±39.96; 10.72±11.27 vs 8.370±8.346 IU/ml, respectively) (Table 3).
Table 2. Comparison autoantibodies between GFD group of CD patients and control.
Table 3. Comparison of autoantibodies according to pre and post GFD regime.
Correlations between serum anti-tTG IgA level and other autoantibodies in patients
To assess the correlations between anti-tTG IgA and other autoimmune antibodies (anti-Pituitary IgG, anti-TPO IgG, anti-Islet IgG, and anti-ganglioside IgG), Spearman's correlation coefficient (rs) was estimated in ND and GFD patients with CD. In ND group, anti-tTG level showed a significant positive correlation with anti-ganglioside (rs=0.2379, p = 0.0399), however no significant correlation was observed with anti-pituitary IgG, anti-TPO IgG, and anti-islet IgG. Meanwhile, in GFD, anti-tTG IgA confirmed the significant positive correlation with anti-ganglioside (rs=0. 2310, p = 0.0461) as well as with anti-pituitary (rs=0. 2796, p = 0.0151). Both anti-TPO and anti-islet showed no correlation with anti-tTG IgA level in GFD group (Table 4).
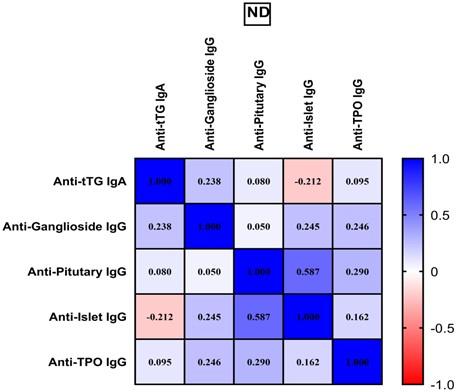
Furthermore, a correlation matrix was created to determine the relationships between all assessed autoantibodies in the ND and GFD groups. Weak correlations were recorded between anti-ganglioside IgG and anti-islet IgG (rs= 0.244, p= 0.034), anti-islet IgG and anti-TPO IgG (rs= 0.252, p= 0.028), whereas anti-pituitary IgG was moderately correlated with anti-islet (rs= 0.586, p=0.000) and anti TPO (rs= 0.426, p=0.000) in ND of CD patients (Figure 1).
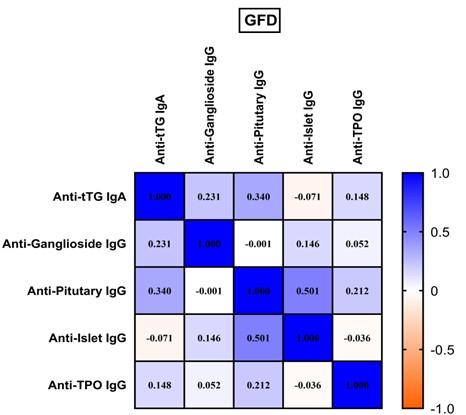
After 4 months of GFD, correlation status was as follows: anti-pituitary IgG was still in moderate correlation with anti-islet IgG (rs= 0.501, p=0.000) and anti TPO IgG (rs= 0.426, p=0.000) while anti-islet IgG maintained weak correlation with anti TPO IgG (rs= 0.252, p= 0.028) (Figure 2).
Table 4. Correlation of anti-tTG IgA and other autoantibodies of ND with GFD patients.
DISCUSSION
It has been demonstrated that a GFD may reduce the signs and symptoms of several autoimmune non-endocrine diseases as well as some underlying endocrine disorders [12]. The present work investigated the influence of gluten on the level of the number of autoantibodies associated with CD-related autoimmune illnesses, including anti-pituitary IgG, anti-TPO-IgG, anti-islet IgG, and anti-ganglioside IgG, and analyzed the correlation between anti-tTG-IgA and the mentioned autoantibodies.
Since consumption of gluten can cause autoimmune disorders, Lerner et al. hypothesized that protein-glutamine-glutamyl-transferases may play a role in the emergence of autoimmune diseases [13]. By enzymatically altering peptides in the gut lumen, endogenous protein-glutamine-glutamyl-transferases released by the gut microbiota have the potential to trigger systemic autoimmunity. These actions resulted in the cross-linking of naïve proteins, which may produce immunogenic neo-epitopes and be the cause of pathogenic autoimmune diseases [14]. Therefore, persistent consumption of dietary gluten in people who are prone to autoimmunity may set off a chain of harmful events that either causes autoimmune illnesses or favors their progression, whereas gluten abstinence may help to halt this chain of events [15].
Different immunological tests including tTG-IgA should be utilized for follow-up care, according to new guidelines on the diagnosis and treatment of CD issued by the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition [16]. Additionally, it correlates with the severity of intestinal harm and villous abnormalities [17]. The detection of positive serology early-onset CD can be facilitated by In-situ analysis of tTG-IgA immune deposits [18]. Due to the fact that most CD patients have symptomatic improvement with treatment, where follow-up is crucial. The results are consistent with earlier research which demonstrated a clear increase in antibody concentration in individuals with managed CD after a gluten challenge [19]. Similar to that, this study observed the levels of antibody drop sharply in the first few months after beginning GFD supports the findings of earlier investigations [20]. Our results came in parallel with a previous study [21] which proved tTG-IgA undergoes remission after months from starting a GFD. The correlation between tTG-IgA and other biomarkers in CD showed significant association before and after adherence to GFD. GFD markedly reduced the level of tTG-IgA [12]. Another study showed that after 1 and 2 years on GFD, a significant number of CD patients were normalized their level of tTG-IgA [22].
According to Shamriz et al. [15], a GFD may have several beneficial impacts on autoimmune illnesses other than CD, including the regulation of the microbiome, reduction of oxidative stress, and changes in epigenetic variables. It was observed that GFD may result in a considerable decrease in anti-pituitary antibody titers and a restoration of anterior pituitary function in individuals with lymphocytic hypophysitis in the preclinical stage and CD, or it may stop the condition from progressing from subclinical to clinically overt [23]. Numerous research has examined how GFD affects thyroid disease in individuals who also have CD. GFD could reverse thyroid problems in certain individuals, according to an Italian multicenter study that examined the thyroid function of 128 ND of CD patients before and one year after the introduction of a GFD [24]. In a recent study by Velija et al [25], participants were had been divided into two groups: those who consumed selenium along with a GFD and those who used selenium supplements alone, without any other dietary changes. At the conclusion of the research, more GFD participants—37/50 (74%), compared to 28/48 (58.3%) of the control group—had reached euthyroid status, suggesting that a prolonged GFD could induce progressive disappearance of anti-pituitary antibody with recovery of normal pituitary function when previously impaired. The GFD group experienced a greater drop in serum anti-TPO (by 49%) than the control group (34%). Both groups experienced a decline in thyroid-stimulating hormone (TSH) and anti-tTG levels, although gluten exclusion increased anti-TPO levels, antibodies are specific diagnostic marker for Hashimoto thyroiditis [26]. According to Krysiak and his collagenase's findings, women with autoimmune thyroid illness may benefit clinically from the GFD [27]. These results come in parallel with present study which indicated the decreasing of anti-pituitary IgG and anti-TPO IgG at significant level only during 4 months of treatment. This is plausible given the ability of GFD to restore pituitary cells by preventing the gluten immunogenic activity, inhibiting vascular hyperpermeability, and blocking cytokines—all factors that initiate and sustain the autoimmune inflammatory pituitary process with the potential for remission [23].
Islet autoantibody testing is primarily used to distinguish T1DM from diabetes caused by other non-autoimmune issues [28]. Autoimmune diseases are very common in people with CD. Additionally, it has been discovered that the prevalence of organ-specific autoantibodies like islet-specific autoimmunity is higher than expected in CD patients who are untreated [29]. Depending on reference values, all ND patients were detected positive for islet antibodies and diagnosed with T1DM [30]. Most studies revealed that the prevalence of islet-specific autoimmune antibodies increased in CD, besides these specific antibodies appear to be dependent on gluten and frequently vanished after a GFD [31]. Various research showed that GFD highly affect the anti-islet antibodies level especially in CD patients diagnosed with T1DM [32]. At the time of diagnosis, around 6% of people with CD have at least one of the primary pancreatic islet autoantibodies that define T1DM [33]. The question of whether GFD could lessen the islet-specific autoimmunity identified in patients upon CD diagnosis is one that has received little research. In 71% of CD patients, GFD (median duration 39 months) was linked to a decline or absence of the islet-specific autoantibodies. At CD diagnosis, only one of the islet-specific autoimmune markers was positive in over 80% of the patients who went on to become autoantibody-negative during the GFD, and none of them went on to acquire diabetes [34]. The findings of this study revealed that in all of examined patients, islet-specific autoantibodies appeared to be gluten-dependent and tended to diminish after a GFD. Moreover, Hansen et al. [35] cited a GFD has a preventive effect on T1DM via modifying the systemic immune system and lowers autoimmune inflammation in salivary glands and the pancreas in NOD mice. When both illnesses are present in the same patient, T1D typically begins before CD and only about 5% of people with T1D will also have CD [36]. Human lymphocyte antigen (HLA) genotypes (DR-DQ) that are shared by both of these illnesses have historically been thought to explain their co-occurrence. However, data indicates that non-genetic or environmental factors may have a significant impact on the connection between these illnesses [37].
Anti-ganglioside was distinguished as antibodies reacted with cerebellar cells. Autoimmune neuropathies which contain antiganglioside antibodies that respond to self-gangliosides [38]. Many studies denoted the association of CD and anti-ganglioside antibodies, however in most cases the secretion of anti-ganglioside is highly correlated with neurological dysfunction in CD patients [39, 40]. In CD patients, gluten-sensitive enteropathy has been connected to sleep and neurological issues. However, much research described the presence of these antibodies in patients with CD, especially those with neuropathy disorders [41]. Anti-ganglioside antibodies and neurological symptoms were observed to disappear in roughly half of patients after GFD [42]. In 64% of patients with CD and neurological symptoms, anti-ganglioside antibodies were detected, and 50% of these individuals had blood levels that were undetectable after a year of following GFD [43]. The same result was appeared in our study, anti-ganglioside IgG declined after abstracting gluten eating for 4 months. Anti-ganglioside IgG are especially important in systemic immune responses that start in the gut mucosa. Hadjivassiliou et al referred to there are clinical indications of the autoimmune reaction against the neural anti-transglutaminases may primarily impact the brain or the peripheral nervous system, with minimal stomach involvement [44]. Experimental data points to the possibility of antibody cross-reactivity between gluten peptides and antigenic epitopes on Purkinje cells [45]. Moreover, cross-reactivity with Purkinje cell epitopes is seen in serum from CD patients without neurological symptoms [44].
The elevated level of tTG-IgA in CD patients was confirmed with the elevation of anti-ganglioside level in positive correlation. For this reason, adherence to GFD for more than 1 year markedly decreased the level of tTG-IgA which in consequence dropped the level of anti-ganglioside. On another hand, many reports proved that the concentration of antiganglioside antibodies was not dependent on adherence to GFD [46]. While a study suggested that the processes for producing these antibodies don't depend on consuming gluten [47]. Even without intestinal abnormalities, circulating. anti-ganglioside and tTG may be found in patients with neurological symptoms [48] due to the expression of transglutaminase 6 in the central nervous system by activated astrocytes, microglia, and neurons and is regarded as an autoantigen and the first sign of neurological involvement in gluten-related illnesses, particularly in gluten ataxia [49]. Anti-pituitary antibodies were observed to developed in CD patients especially those with pituitary gland disorders. Many studies revealed the correlation of CD and anti-pituitary antibodies , in addition, strict adherence to GFD showed a significant drop in tTG-IgA and anti-pituitary antibodies which enable the CD patients with short stature to catch-up growth after 1 to 2 years strict GFD [50], these results have been reinforced by the resulting significant positive correlation between tTG-IgA and anti-pituitary antibodies levels despite of the short duration treatment with GFD regime.
CONCLUSIONS
In addition to the fact that abstaining from eating food containing gluten, which is the main treatment of CD. The present study concluded that gluten free meals have a significant impact on many autoimmune disorders by affecting the disease severity depending on the reduction in the levels of autoantibodies specific for diagnosis of each disease. Therefore, it's critical to identify autoantibodies that have developed against the neurological system's and endocrine system's organs both at the start of CD and in follow up cases.
ACKNOWLEDGEMENT
None
AUTHOR CONTRIBUTIONS
NJAB participated in the main conception and design of the experiments. ZNE contributed to perform the lab work, collect samples, analyze data and draft the article. NJAB revised it critically for important intellectual content. The final approval of the version to be published was made by both authors.
CONFLICTS OF INTEREST
There is no conflict of interest among the authors.
References
- [1]Mohta S, Rajput MS, et al. Emergence of celiac disease and gluten-related disorders in asia. Journal of neurogastroenterology and motility. 2021;27:337-46.
- [2]Choung RS, Larson SA, et al. Prevalence and morbidity of undiagnosed celiac disease from a community-based study. Gastroenterology. 2017;152:830-9.e5.
- [3]Majsiak E, Choina M, et al. Clinical manifestation and diagnostic process of celiac disease in poland-comparison of pediatric and adult patients in retrospective study. Nutrients. 2022;14.
- [4]Caio G, Volta U, et al. Celiac disease: A comprehensive current review. BMC medicine. 2019;17:1-20.
- [5]Husby S, Koletzko S, et al. European society for pediatric gastroenterology, hepatology, and nutrition guidelines for the diagnosis of coeliac disease. Journal of pediatric gastroenterology and nutrition. 2012;54:136-60.
- [6]Verdu EF, Danska JS. Common ground: Shared risk factors for type 1 diabetes and celiac disease. Nature immunology. 2018;19:685-95.
- [7]Basu M, Pandit K, et al. Profile of auto-antibodies (disease related and other) in children with type 1 diabetes. Indian Journal of Endocrinology and Metabolism. 2020;24:256.
- [8]Shee A, Prior S, et al. Can islets autoantibody profile at diagnosis of type 1 diabetes in children and adolescents predict the future development of hashimoto’s thyroiditis and coeliac disease? Journal of Pediatric Endocrinology. 2020;5:1-5.
- [9]Naiyer AJ, Shah J, et al. Tissue transglutaminase antibodies in individuals with celiac disease bind to thyroid follicles and extracellular matrix and may contribute to thyroid dysfunction. Thyroid. 2008;18:1171-8.
- [10]Lerner A, Jeremias P, et al. Gut-thyroid axis and celiac disease. Endocrine connections. 2017;6:R52-R8.
- [11]Kass-Iliyya L, Sarrigiannis PG, et al. Glycine receptor antibodies and coeliac disease-related neurological dysfunction. Cerebellum & Ataxias. 2021;8:1-4.
- [12]Iervasi E, Auricchio R, et al. Serum il-21 levels from celiac disease patients correlates with anti-ttg iga autoantibodies and mucosal damage. Autoimmunity. 2020;53:225-30.
- [13]Lerner A, Jeremias P, et al. The world incidence of celiac disease is increasing: A review. Int J Recent Sci Res. 2015;7:5491-6.
- [14]Lerner A, Aminov R, et al. Transglutaminases in dysbiosis as potential environmental drivers of autoimmunity. Frontiers in microbiology. 2017;8:66.
- [15]Shamriz O, Mizrahi H, et al. Microbiota at the crossroads of autoimmunity. Autoimmunity reviews. 2016;15:859-69.
- [16]Alshebani AMH, Abdalhamza ZSJRLdH. The role of celiac disease antibodies in the follow up of patient on gluten free diet. 2018;13:561-6.
- [17]Singh A, Pramanik A, et al. Non-invasive biomarkers for celiac disease. Journal of Clinical Medicine. 2019;8:885.
- [18]Lal R, Bhardwaj R, et al. Usefulness of a double immunofluorescence technique for detection of intestinal ttg-iga deposits in diabetic and non-diabetic children with celiac disease. Pediatrics & Neonatology. 2022.
- [19]Bonamico M, Nenna R, et al. Radioimmunological detection of anti-transglutaminase autoantibodies in human saliva: A useful test to monitor coeliac disease follow-up. 2008;28:364-70.
- [20]Leffler DA, Edwards George JB, et al. A prospective comparative study of five measures of gluten-free diet adherence in adults with coeliac disease. Alimentary pharmacology & therapeutics. 2007;26:1227-35.
- [21]Sbravati F, Cosentino A, et al. Antitissue transglutaminase antibodies’ normalization after starting a gluten-free diet in a large population of celiac children-a real-life experience. Digestive and Liver Disease. 2022;54:336-42.
- [22]Giner-Pérez L, Donat E, et al. Study of the immune response in celiac patients with selective iga deficiency who start a gluten-free diet. Clinical and Experimental Medicine. 2023.
- [23]Bellastella G, Maiorino MI, et al. Remission of pituitary autoimmunity induced by gluten-free diet in patients with celiac disease. The Journal of Clinical Endocrinology & Metabolism. 2020;105:2252-61.
- [24]Ashok T, Patni N, et al. Celiac disease and autoimmune thyroid disease: The two peas in a pod. 2022;14.
- [25]Velija AZ, Hadzovic-Dzuvo A, et al. The effect of selenium supplementation and gluten-free diet in patients with subclinical hypothyroidism affected by autoimmune thyroiditis. Endocrine Abstracts: Bioscientifica; 2020.
- [26]Chaudhuri J, Mukherjee A, et al. Hashimoto’s encephalopathy: Case series and literature review. Current Neurology and Neuroscience Reports. 2023;23:167-75.
- [27]Krysiak R, Szkróbka W, et al. The effect of gluten-free diet on thyroid autoimmunity in drug-naïve women with hashimoto's thyroiditis: A pilot study. Exp Clin Endocrinol Diabetes. 2019;127:417-22.
- [28]Bonifacio E, Achenbach P. Birth and coming of age of islet autoantibodies. Clinical and experimental immunology. 2019;198:294-305.
- [29]Kahaly GJ, Frommer L, et al. Celiac disease and glandular autoimmunity. 2018;10:814.
- [30]Tang W, Liang H, et al. Diagnostic value of combined islet antigen-reactive t cells and autoantibodies assays for type 1 diabetes mellitus. Journal of diabetes investigation. 2021;12:963-9.
- [31]Pham-Short A, Donaghue KC, et al. Quality of life in type 1 diabetes and celiac disease: Role of the gluten-free diet. The Journal of Pediatrics. 2016;179:131-8.e1.
- [32]Eland I, Klieverik L, et al. Gluten-free diet in co-existent celiac disease and type 1 diabetes mellitus: Is it detrimental or beneficial to glycemic control, vascular complications, and quality of life? 2023;15:199.
- [33]Cohn A, Sofia AM, et al. Type 1 diabetes and celiac disease: Clinical overlap and new insights into disease pathogenesis. Current diabetes reports. 2014;14:517.
- [34]Tiberti C, Montuori M, et al. Gluten-free diet impact on dynamics of pancreatic islet-specific autoimmunity detected at celiac disease diagnosis. 2020;21:774-80.
- [35]Hansen CH, Larsen CS, et al. Gluten-free diet reduces autoimmune diabetes mellitus in mice across multiple generations in a microbiota-independent manner. Journal of Autoimmunity. 2022;127:102795.
- [36]Pham-Short A, Donaghue KC, et al. Screening for celiac disease in type 1 diabetes: A systematic review. Pediatrics. 2015;136:e170-e6.
- [37]Hagopian W, Lee H-S, et al. Co-occurrence of type 1 diabetes and celiac disease autoimmunity. Pediatrics. 2017;140.
- [38]Cutillo G, Saariaho A-H, et al. Physiology of gangliosides and the role of antiganglioside antibodies in human diseases. Cellular & Molecular Immunology. 2020;17:313-22.
- [39]Granito A, Tovoli F, et al. Anti-ganglioside antibodies and celiac disease. Allergy, Asthma & Clinical Immunology. 2021;17:53.
- [40]Volta U, De Giorgio R, et al. Anti-ganglioside antibodies in coeliac disease with neurological disorders. Digestive and liver disease : official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver. 2006;38:183-7.
- [41]Güngör S, Celiloglu ÖS, et al. Frequency of celiac disease in attention-deficit/hyperactivity disorder. Journal of Pediatric Gastroenterology and Nutrition. 2013;56.
- [42]Suzer Gamli I, Keceli Basaran M. The effect of a gluten-free diet on sleep disturbances in children with celiac disease. Nature and Science of Sleep. 2022;14:449-56.
- [43]Alhosain AI, Alshammari GM, et al. Long-term effect of gluten-free diets on nutritional status, body composition, and associated factors in adult saudi females with celiac disease. 2022;14:2090.
- [44]Hadjivassiliou M, Grünewald R, et al. Neuropathy associated with gluten sensitivity. Journal of Neurology, Neurosurgery & Psychiatry. 2006;77:1262-6.
- [45]Hadjivassiliou M, Boscolo S, et al. The humoral response in the pathogenesis of gluten ataxia. Neurology. 2002;58:1221-6.
- [46]Przybylska-Feluś M, Zwolińska-Wcisło M, et al. Concentrations of antiganglioside m1 antibodies, neuron-specific enolase, and interleukin 10 as potential markers of autonomic nervous system impairment in celiac disease. 2016;126.
- [47]Briani C, Zara G, et al. Neurological complications of celiac disease and autoimmune mechanisms: A prospective study. Journal of neuroimmunology. 2008;195:171-5.
- [48]Schuppan D, Junker Y, et al. Celiac disease: From pathogenesis to novel therapies. Gastroenterology. 2009;137:1912-33.
- [49]Hadjivassiliou M, Croall ID, et al. Neurologic deficits in patients with newly diagnosed celiac disease are frequent and linked with autoimmunity to transglutaminase 6. Clinical Gastroenterology and Hepatology. 2019;17:2678-86. e2.
- [50]Walia R, Singh A, et al. Look beyond gluten in short stature with celiac disease – a prospective, interventional study. Indian Journal of Pediatrics. 2021;88:550-4.